

Investor Presentation – June 2022 Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. Filed Pursuant to Rule 433 of the Securities Act of 1933 Issuer Free Writing Prospectus dated June 21, 2022 Relating to Preliminary Prospectus dated June 21, 2022 Registration No. 333 - 263694

2 This document contains forward - looking statements. In addition, from time to time, we or our representatives may make forward - lo oking statements orally or in writing. We base these forward - looking statements on our expectations and projections about future events, which we derive from the information curren tly available to us. Such forward - looking statements relate to future events or our future performance, including: our financial performance and projections; our growth in revenue and ear nings; and our business prospects and opportunities. You can identify forward - looking statements by those that are not historical in nature, particularly those that use terminology such as “may,” “should,” “expects,” “anticipates,” “contemplates,” “estimates,” “believes,” “plans,” “projected,” “predicts,” “potential,” or “hopes” or the negative of these or similar terms . F actors that may cause actual results to differ materially from current expectations include, among other things, those listed under the heading “Risk Factors” and elsewhere in the registration sta tem ent that we have filed with the U.S. Securities and Exchange Commission. Forward - looking statements are only predictions. The forward - looking events discussed in this document and other sta tements made from time to time by us or our representatives, may not occur, and actual events and results may differ materially and are subject to risks, uncertainties a nd assumptions about us. We are not obligated to publicly update or revise any forward - looking statement, whether as a result of uncertainties and assumptions, the forward - looking events discu ssed in this document and other statements made from time to time by us or our representatives might not occur. Past performance is not indicative of future results. There is now guar ant ee that any specific outcome will be achieved. Investments may be speculative, illiquid and there is a total risk of loss. Free Writing Prospectus We have filed a registration statement (including a preliminary prospectus) with the SEC for the offering to which this commu nic ation relates. The registration statement has not yet become effective. Before you invest, you should read the preliminary prospectus in that registration statement (including the risk fac tors described therein) and other documents that we have filed with the SEC for more complete information about us and this offering. You may access these documents for free by visiting EDGAR on the SEC Website at http://www.sec.gov. The preliminary prospectu s w as filed January 25, 2022 and is available on the SEC website at http://www.sec.gov. Alternatively, VRAX and any underwriter or dealer participating in the offering will arrange to send you the prospectus if you request it by calling Boustead Securities, LLC at 949.502.4408 or by email at offerings@boustead1828.com or standard mail at Boustead Securities, LLC, Attn: Eq uity Capital Markets, 6 Venture, Suite 395, Irvine, CA 92618, USA. Form CRS/Reg BI Disclaimer: Boustead Securities, LLC is registered with the Securities and Exchange Commission (SEC) as a broker - dealer and is a member of t he Financial Industry Regulatory Authority (FINRA) and the Securities Investor Protection Corporation (SIPC). Brokerage and investment advisory services and fees differ and it is impor tan t for you to understand these differences. Free and simple tools are available to research firms and financial professionals at Investor.gov/CRS, which also provides educational materi als about broker - dealers, investment advisers, and investing. When we provide you with a recommendation, we have to act in your best interest and not put our interest ahead of yours. At t he same time, the way we make money creates a conflict with your interests. Please strive to understand and ask us about these conflicts because they can affect the recommendations we p rov ide you. There are many risks involved with investing. For Boustead Securities customers and clients, please see our Regulation Best Interest Relationship Guide on the Form CRS Reg BI pag e on our website at https://www.boustead1828.com/form - crs - reg - bi. For FlashFunders ’ visitors, you may review the Form CRS of Boustead Securities under the Form CRS section. Please also carefully review and verify the accuracy of the information you provide us on account applications, subscription documents and others. © 2022 Disclaimers Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.

3 We have limited operating history, have incurred operating losses for the six months ended September 30, 2021 and the years ended March 31, 2021 and 2020 and expect to incur significant losses for the foreseeable future. We may not generate sufficient revenue or become profitable or, if we achieve profitability, we may not be able to sustain it. Therefore, it is too early to draw meaningful conclusions from the financial performance of the Group due to the change in our business focus since 2020 as our commercialized brands are ViraxClear and ViraxCare , which have been put into market since 2020. We expect to make significant investments with respect to our gross income in our continued research and development of new products and services, which may not be successful. If we are not successful in leveraging Virax Immune platform to discover, develop and commercialize additional products and services, our ability to expand our business and achieve our strategic objectives would be impaired. Our efforts to develop T - Cell In - Vitro Diagnostic Test may not be successful, and it may not yield the insights we expect at all or on a timetable that allows us to develop or commercialize any new diagnostic products. If we are not successful in obtaining regulatory approvals for our Virax Immune products, we may not be able to commercialize our products in the expected timeframe or at all, and our ability to expand our business and achieve our strategic objectives would be impaired. We will face significant challenges in successfully commercializing our products, particularly in new markets. Our business, financial condition and results of operations will depend on the market acceptance and increased demand of our products by hospitals, governments and public health departments, as well as physicians others in the medical community, and the growing proportion of the population who are interested in taking personal charge over their health and wellbeing. The success of some of our products partially depends on the continued demand for diagnostic and products linked to COVID - 19 and other major viral diseases. Risk Factors Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. The success of our proprietary technology T - Cell testing requires us to proceed through clinical and validation studies successfully which is not guaranteed. New market opportunities may not develop as quickly as we expect, limiting our ability to market and sell our products successfully. We do not have in place any supply contracts with two of our key suppliers, and any disruptions from such key suppliers could adversely affect our business and results of operations. We rely on a limited number of suppliers or, in many cases, single suppliers, for laboratory equipment and materials and may not be able to find replacements or immediately transition to alternative suppliers. Our suppliers may experience development or manufacturing problems or delays that could limit the growth of our revenue or increase our losses. We have a significant customer concentration, with a limited number of customers accounting for a large portion or all of our revenues. The COVID - 19 pandemic could adversely impact portions of our business that rely on research and development activities or clinical trials and delay or disrupt our pipeline, which may adversely impact revenue. Our efforts to discover and develop products and services related to COVID - 19 and major viral threats, namely Virax Immune products, may not be successful from either a platform extension or commercialization perspective. We may be liable for improper collection, use or appropriation of personal information provided by our customers. The in - vitro diagnostics industry is subject to rapid change, which could make our diagnostics platform and related products and services that we develop obsolete. Our business could suffer if we lose the services of, or are unable to attract and retain, key members of our senior management, key advisors or other personnel. We depend on our information technology systems and any failure of these systems could harm our business.

4 We face risks related to natural disasters, health epidemics and other outbreaks, specifically the coronavirus, which could significantly disrupt our operations. If we are not able to adequately protect our proprietary intellectual property and information, and protect against third party claims that we are infringing on their intellectual property rights, our results of operations could be adversely affected. If we are unable to adequately protect our intellectual property rights, or if we are accused of infringing on the intellectual property rights of others, our competitive position could be harmed or we could be required to incur significant expenses to enforce or defend our rights. We intend to apply for patents in the United States, subject to approval from the relevant regulatory bodies. If we do not obtain protection under the Hatch - Waxman Amendments and similar non - U.S. legislation for extending the term of patents covering each of our product candidates, our business may be materially harmed. Intellectual property rights do not necessarily address all potential threats to our competitive advantage and changes in patent laws or patent jurisprudence could diminish the value of patents in general, thereby impairing our ability to protect our products. We enjoy only limited geographical protection with respect to certain patents and may face difficulties in certain jurisdictions, which may diminish the value of intellectual property rights in those jurisdictions. Failure to partially acquire the proprietary technology from a European Union based materials technology company could have an adverse effect on our planned results of operations for our Virax Immune brand and our business. Litigation or other proceedings or third - party claims of intellectual property infringement could require us to spend significant time and money and could prevent us from selling our products or affect our stock price. If we are unable to protect the confidentiality of our trade secrets, our business and competitive position could be harmed. Third parties may assert ownership or commercial rights to inventions we develop, which could have a material adverse effect on our business. Risk Factors (Cont.) Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. Third parties may assert that our employees or contractors have wrongfully used or disclosed confidential information or misappropriated trade secrets, which could result in litigation. The regulatory environment for IVD could change, resulting a new procedure for achieving approvals for various global marketplaces which might adversely affect Virax’s ability to enter various markets. If we fail to comply with extensive regulations of domestic and international regulatory authorities, sales of our products in new markets and the development and commercialization of any new product candidates could be delayed or prevented. If we or our suppliers fail to comply with ongoing regulatory requirements, or if we experience unanticipated problems with our products, these products could be subject to restrictions or withdrawal from the market. We may potentially be subject to product liability claims. Our inadvertent or unintentional failure to comply with complex government regulations concerning privacy of medical and personal information could subject us to fines and adversely affect our reputation. A disruption in our computer networks, including those related to cybersecurity, could adversely affect our financial performance. We are subject to the U.K. Bribery Act and other anti - corruption laws, as well as export control laws, customs laws, sanctions laws and other laws governing our operations. If we fail to comply with these laws, we could be subject to civil or criminal penalties, other remedial measures, and legal expenses, which could adversely affect our business, results of operations and financial condition. Recent developments relating to the United Kingdom’s withdrawal from the European Union could adversely affect us. We are exposed to unanticipated changes in tax laws and regulations, adjustments to our tax provisions, exposure to additional tax liabilities, or forfeiture of our tax assets.

5 We may rely on dividends and other distributions on equity paid by our subsidiaries to fund any cash and financing requirements we may have, and any limitation on the ability of our subsidiaries to make payments to us could have a material adverse effect on our ability to conduct our business. Developments in the social, political, regulatory and economic environment in the countries where we operate, may have a material and adverse impact on us. Disruptions in the international trading environment may seriously decrease our international sales. A downturn in the Hong Kong, China or global economy, and economic and political policies of China could materially and adversely affect our business and financial condition. The Hong Kong legal system embodies uncertainties which could limit the legal protections available to us. Uncertainties with respect to the PRC legal system, including uncertainties regarding the enforcement of laws, and sudden or unexpected changes in laws and regulations in China could adversely affect us. The Chinese government exerts substantial influence over the manner in which we must conduct our business activities, and may intervene or influence our operations at any time, or may exert more oversight and control over offerings conducted overseas, which could significantly limit or completely hinder our ability to offer or continue to offer our Ordinary Shares to investors and could cause the value of our Ordinary Shares to significantly decline or become worthless. Enhanced scrutiny over acquisition transactions by the PRC tax authorities may have a negative impact on potential acquisitions we may pursue in the future. The approval of the China Securities Regulatory Commission and other PRC governmental authorities are not required in connection with this offering, and, if required, we cannot predict whether we will be able to obtain such approval. Risk Factors (Cont.) Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. The Chinese government may exercise significant oversight and discretion over the conduct of Shanghai Xitu’s business and may intervene in or influence its operations at any time, which could result in a material change in its operations and/or the value of our securities. Changes in China’s economic, political or social conditions or government policies could have a material adverse effect on our Company’s business and results of operations we may pursue in the future. PRC regulation of loans to and direct investment in PRC entities by offshore holding companies and governmental control of currency conversion may delay us from using part of the proceeds of this offering to make loans or additional capital contributions to our PRC subsidiary, which could materially and adversely affect our liquidity and our ability to fund and expand our business. Restrictions on currency exchange may limit our ability to utilize our revenues effectively. Dividends paid to our foreign investors and gains on the sale of the Ordinary Shares by our foreign investors may become subject to PRC tax. There has been no prior public market for our Ordinary Shares and an active trading market may never develop or be sustained. The offering price of the primary offering and resale offering could differ. The Resale by the Selling Shareholders may cause the market price of our Ordinary Shares to decline. Our share is expected to initially trade under $5.00 per ordinary share and thus could be known as a penny stock, subject to certain exceptions. Trading in penny stocks has certain restrictions and these restrictions could negatively affect the price and liquidity of our ordinary shares. Our share price may be volatile and may fluctuate. If we fail to meet applicable listing requirements, Nasdaq may delist our Ordinary Shares from trading, in which case the liquidity and market price of our Ordinary Shares could decline.

6 We do not intend to pay cash dividends on our Ordinary Shares in the foreseeable future. We are an emerging growth company within the meaning of the Securities Act and may take advantage of certain reduced reporting requirements. We qualify as a foreign private issuer and, as a result, we will not be subject to U.S. proxy rules and will be subject to Exchange Act reporting obligations that permit less detailed and less frequent reporting than that of a U.S. domestic public company. As a foreign private issuer, we are permitted to adopt certain home country practices in relation to corporate governance matters that differ significantly from Nasdaq corporate governance listing standards. These practices may afford less protection to shareholders than they would enjoy if we complied fully with corporate governance listing standards. There can be no assurance that we will not be a passive foreign investment company, or PFIC, for U.S. federal income tax purposes for any taxable year, which could result in adverse U.S. federal income tax consequences to U.S. holders of our Ordinary Shares. We may lose our foreign private issuer status in the future, which could result in significant additional costs and expenses. Our Ordinary Shares may be prohibited from being traded on a national exchange under the Holding Foreign Companies Accountable Act if the PCAOB is unable to inspect our auditors for three consecutive years beginning in 2021. The delisting of our Ordinary Shares, or the threat of their being delisted, may materially and adversely affect the value of your investment. Our registered public accounting firm, BF Borgers CPA PC, is not headquartered in mainland China or Hong Kong and was not identified in the PACOB’s Determination Report on December 16, 2021 as a firm subject to the PCAOB’s determination. The recent joint statement by the SEC and PCAOB, proposed rule changes submitted by Nasdaq, and the Holding Foreign Companies Accountable Act all call for additional and more stringent criteria to be applied to emerging market companies upon assessing the qualification of their auditors, especially the non - U.S. auditors who are not inspected by the PCAOB. These developments could add uncertainties to our offering. Risk Factors (Cont.) Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. We will have broad discretion in the use of proceeds of this offering designated for working capital and general corporate purposes. Our pre - IPO shareholders will be able to sell their shares after the completion of this offering subject to restrictions under Rule 144 under the Securities Act, which could impact the trading price of our Ordinary Shares. Failure to comply with anticorruption and anti - money laundering laws, including the FCPA and similar laws associated with activities outside of the United States, could subject us to penalties and other adverse consequences. We expect to incur significant additional costs as a result of being a public company, which may materially and adversely affect our business, financial condition and results of operations. Securities analysts may not publish favorable research or reports about our business or may publish no information at all, which could cause our stock price or trading volume to decline. Recently introduced economic substance legislation of the Cayman Islands may impact us and our operations. Because we are incorporated under the laws of the Cayman Islands, our executive office is located in United Kingdom and all of our executive officers and directors are located outside the United States, you may face difficulties in protecting your interests, and your ability to protect your rights through the U.S. Federal or state courts may be limited.
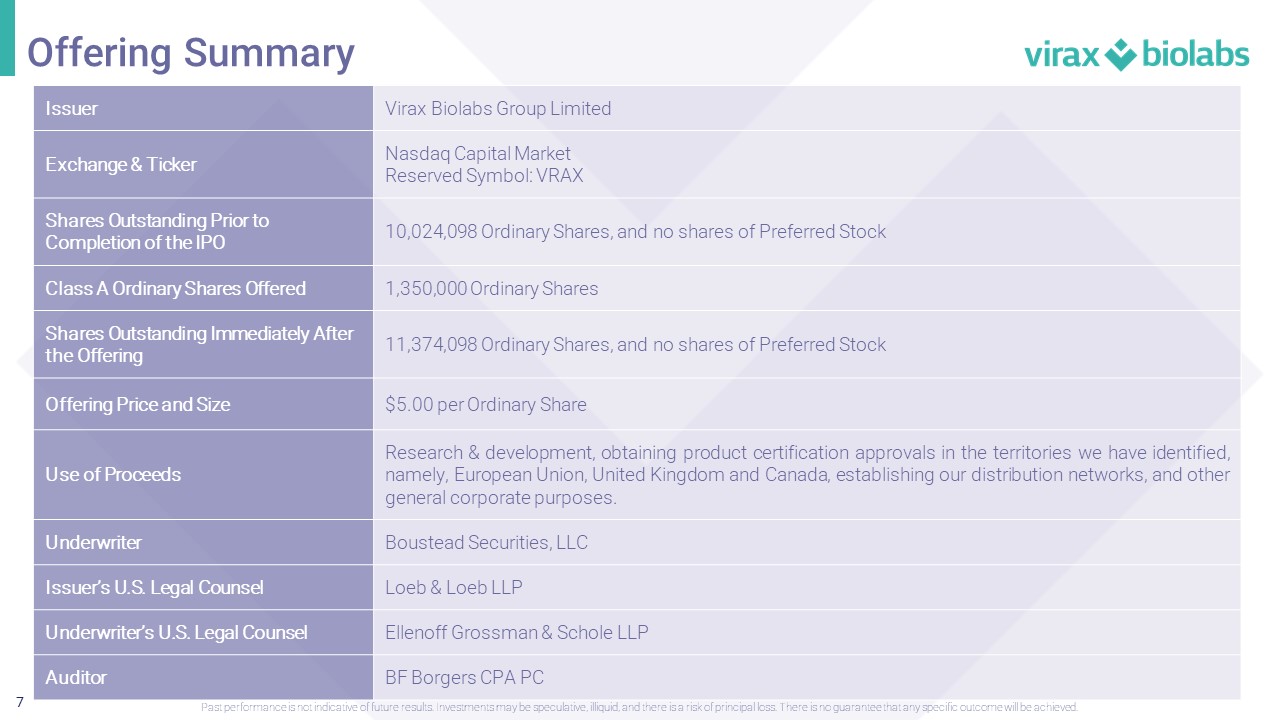
7 Issuer Virax Biolabs Group Limited Exchange & Ticker Nasdaq Capital Market Reserved Symbol : VRAX Shares Outstanding Prior to Completion of the IPO 10 , 024 , 098 Ordinary Shares, and no shares of Preferred Stock Class A Ordinary Shares Offered 1 , 350 , 000 Ordinary Shares Shares Outstanding Immediately After the Offering 11 , 374 , 098 Ordinary Shares, and no shares of Preferred Stock Offering Price and Size $ 5 . 00 per Ordinary Share Use of Proceeds Research & development, obtaining product certification approvals in the territories we have identified, namely, European Union, United Kingdom and Canada, establishing our distribution networks, and other general corporate purposes . Underwriter Boustead Securities, LLC Issuer’s U.S. Legal Counsel Loeb & Loeb LLP Underwriter’s U.S. Legal Counsel Ellenoff Grossman & Schole LLP Auditor BF Borgers CPA PC Offering Summary Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.

Overview 8 Virax Biolabs is an innovative biotechnology company focused on the detection and diagnosis of viral diseases. Virax Biolabs believes it is developing a game - changing immunology platform based around our proprietary T - Cell testing technology, which is particularly effective in the diagnosis of COVID - 19, Hepatitis B, Epstein - Barr virus , Human Papilloma Virus, and other viral threats. Existing revenue streams from ViraxClear and ViraxCare complement the anticipated T - Cell immunology platform via B2B2C sales channels, distribution networks, and an online sales platform. Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.
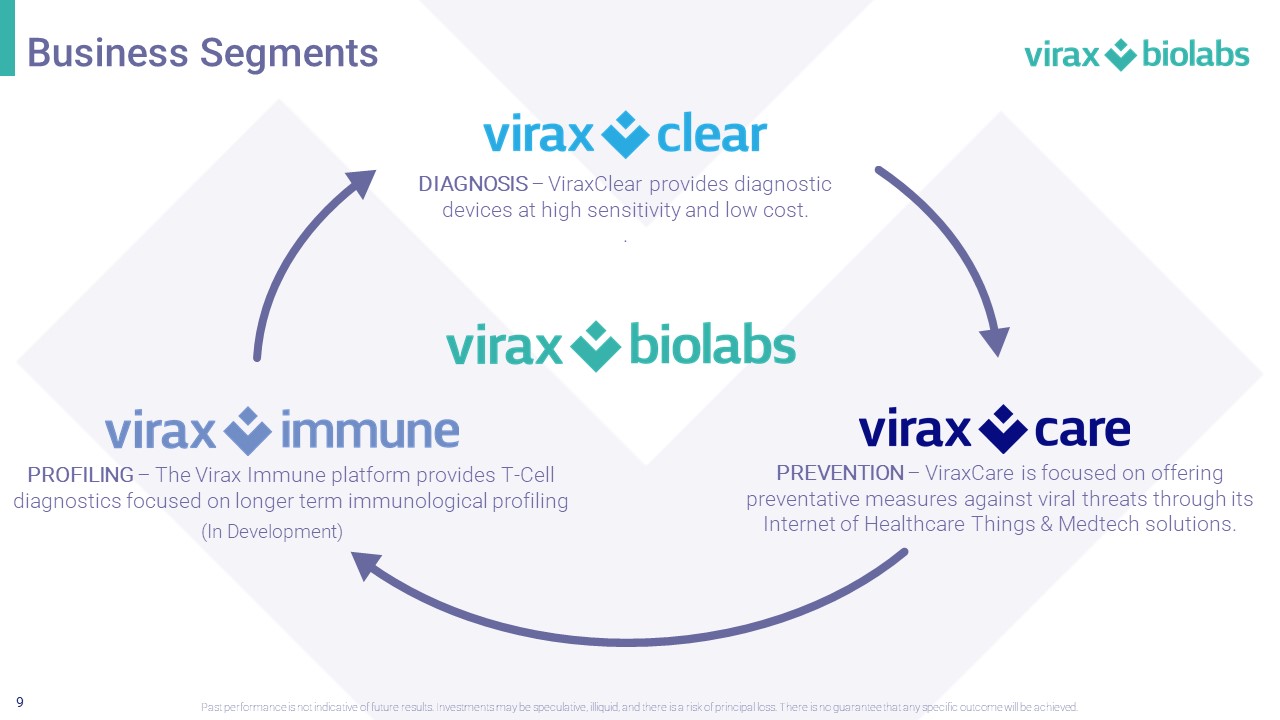
9 PREVENTION – ViraxCare is focused on offering preventative measures against viral threats through its Internet of Healthcare Things & Medtech solutions. PROFILING – The Virax Immune platform provides T - Cell diagnostics focused on longer term immunological profiling DIAGNOSIS – ViraxClear provides diagnostic devices at high sensitivity and low cost. . Business Segments Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. (In Development)
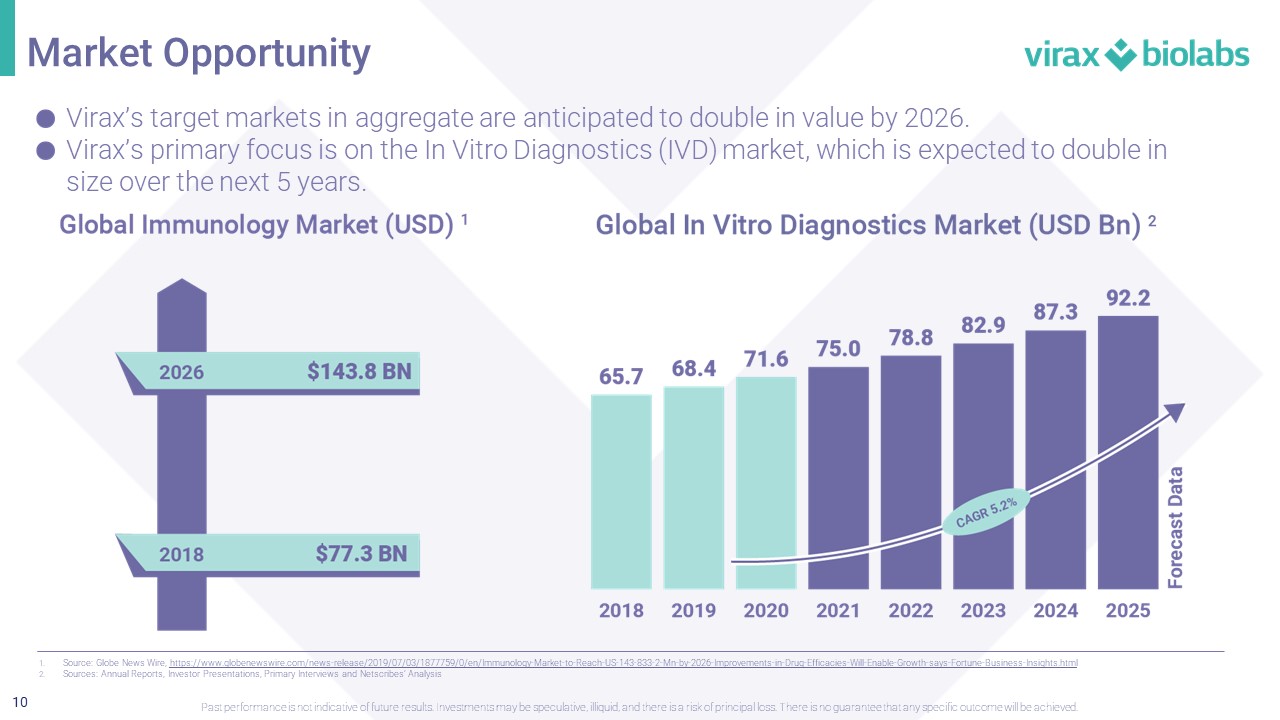
10 Virax’s target markets in aggregate are anticipated to double in value by 2026. Virax’s primary focus is on the In Vitro Diagnostics (IVD) market, which is expected to double in size over the next 5 years. 1. Source: Globe News Wire, https://www.globenewswire.com/news - release/2019/07/03/1877759/0/en/Immunology - Market - to - Reach - US - 143 - 833 - 2 - Mn - by - 2026 - Improvemen ts - in - Drug - Efficacies - Will - Enable - Growth - says - Fortune - Business - Insights.html 2. Sources: Annual Reports, Investor Presentations, Primary Interviews and Netscribes ’ Analysis Market Opportunity Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.
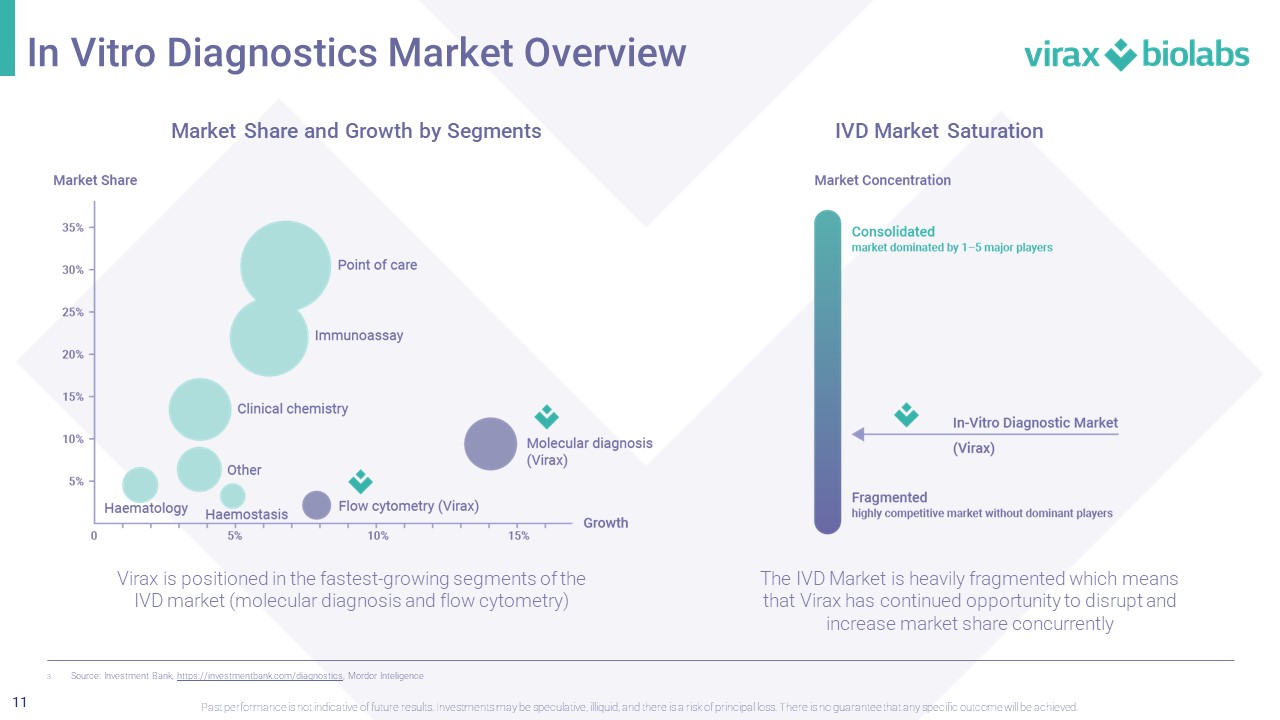
11 IVD Market Saturation In Vitro Diagnostics Market Overview Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. 3. Source: Investment Bank, https://investmentbank.com/diagnostics , Mordor Inteligence Market Share and Growth by Segments Virax is positioned in the fastest - growing segments of the IVD market (molecular diagnosis and flow cytometry) The IVD Market is heavily fragmented which means that Virax has continued opportunity to disrupt and increase market share concurrently

12 TEST PRODUCT APPROVAL REGIONAL APPROVALS SALES/REVENUE ViraxClear Covid - 19 Rapid Antibody IgC /IgM Test — Approved in Europe, South America, Southeast Asia — Revenue generating since 2020 ViraxClear Covid - 19 Antigen Test — Approved in Europe, South America, Southeast Asia — Revenue generating since 2020 — Health Canada & MHRA UK approvals pending Q3 2022 ViraxClear Covid - 19 PCR Rapid Test — Approved in Europe, South America, Southeast Asia — Revenue generating since 2020 ViraxClear Covid - 19 Neutralising Antibody Test — CE Approved — Pending Health Canada & MHRA UK approvals Q2 2022 — Commencing distribution in Q2 2022 ViraxClear – Products Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.
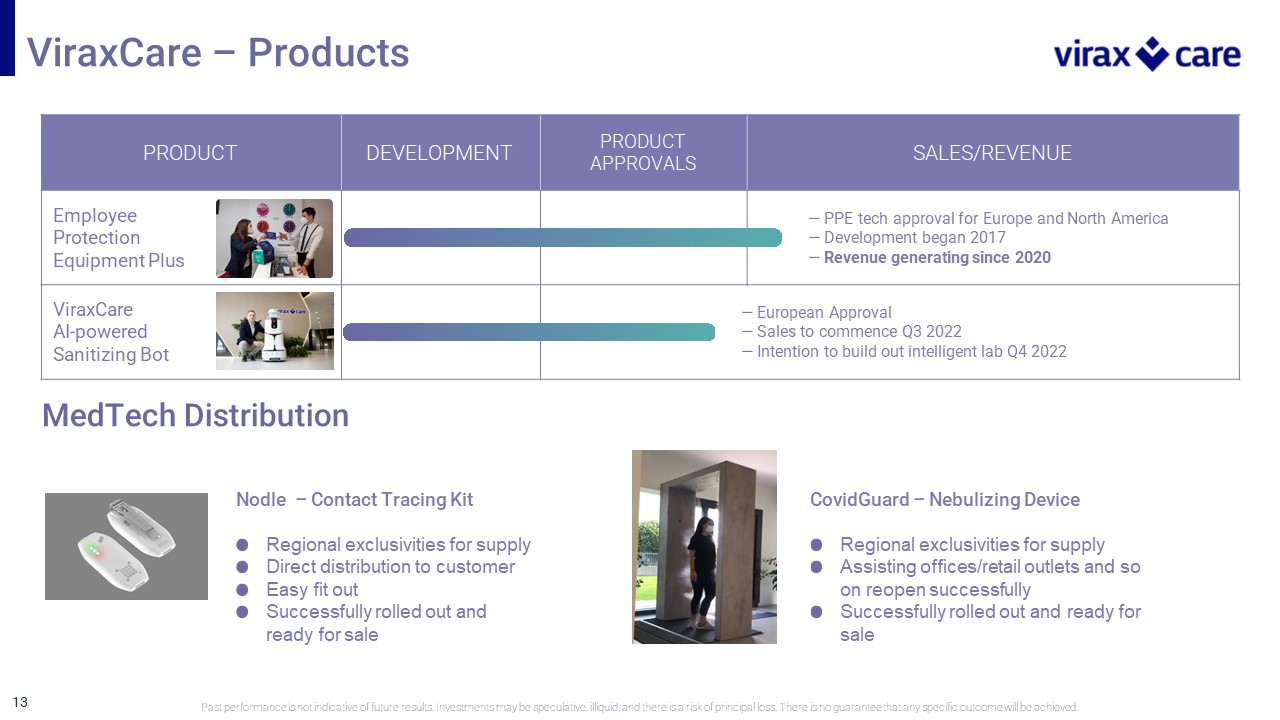
13 PRODUCT DEVELOPMENT PRODUCT APPROVALS SALES/REVENUE Employee Protection Equipment Plus — PPE tech approval for Europe and North America — Development began 2017 — Revenue generating since 2020 ViraxCare AI - powered Sanitizing Bot — European Approval — Sales to commence Q3 2022 — Intention to build out intelligent lab Q4 2022 ViraxCare – Products Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. Nodle – Contact Tracing Kit Regional exclusivities for supply Direct distribution to customer Easy fit out Successfully rolled out and ready for sale CovidGuard – Nebulizing Device Regional exclusivities for supply Assisting offices/retail outlets and so on reopen successfully Successfully rolled out and ready for sale MedTech Distribution

14 T - Cells coordinate the immune system against viral attacks, unlike antibodies. T - Cell detection can give an indication of inherent protection to a range of diseases for those yet to be infected. T - Cells have been shown to provide a level of protection against Covid - 19 and its variants that is more robust and long - lasting than a vaccine. T - Cells may be useful to determine the degree of long - term protection against viral threats. 5, 6, 7, 8, 9 5. Welsh, R. M. & Selin, L. K. No one is naive: the significance of heterologous T - cell immunity. Nat. Rev. Immunol. 2, 417 – 426 (20 02). 6. Le Bert, N., Tan, A.T., Kunasegaran , K. et al. SARS - CoV - 2 - specific T cell immunity in cases of COVID - 19 and SARS, and uninfected controls. Nature 584, 457 – 462 (202 0). https://doi.org/10.1038/s41586 - 020 - 2550 - z 7. Wyllie, D., et al. SARS - CoV - 2 responsive T cell numbers and anti - Spike IgG levels are both associated with protection from COVID - 19: A prospective cohort study in keyworkers. medRxiv 2020.11.02.20222778; doi:https :// doi.org /10.1101/2020.11.02.20222778 8. Shrotri , M., et al. T cell response to SARS - CoV - 2 infection in humans: A systematic review PLOS ONE 16(1): e0245532 (2021). https:// doi.org /10.1371/journal.pone.0245532 9. Wang, Z., Yang, X., Zhong, J. et al. Exposure to SARS - CoV - 2 generates T - cell memory in the absence of a detectable viral infecti on. Nat Commun 12, 1724 (2021). https://doi.org/10.1038/s41467 - 021 - 22036 - z In Development Virax Immune: T - Cell Testing Overview Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.
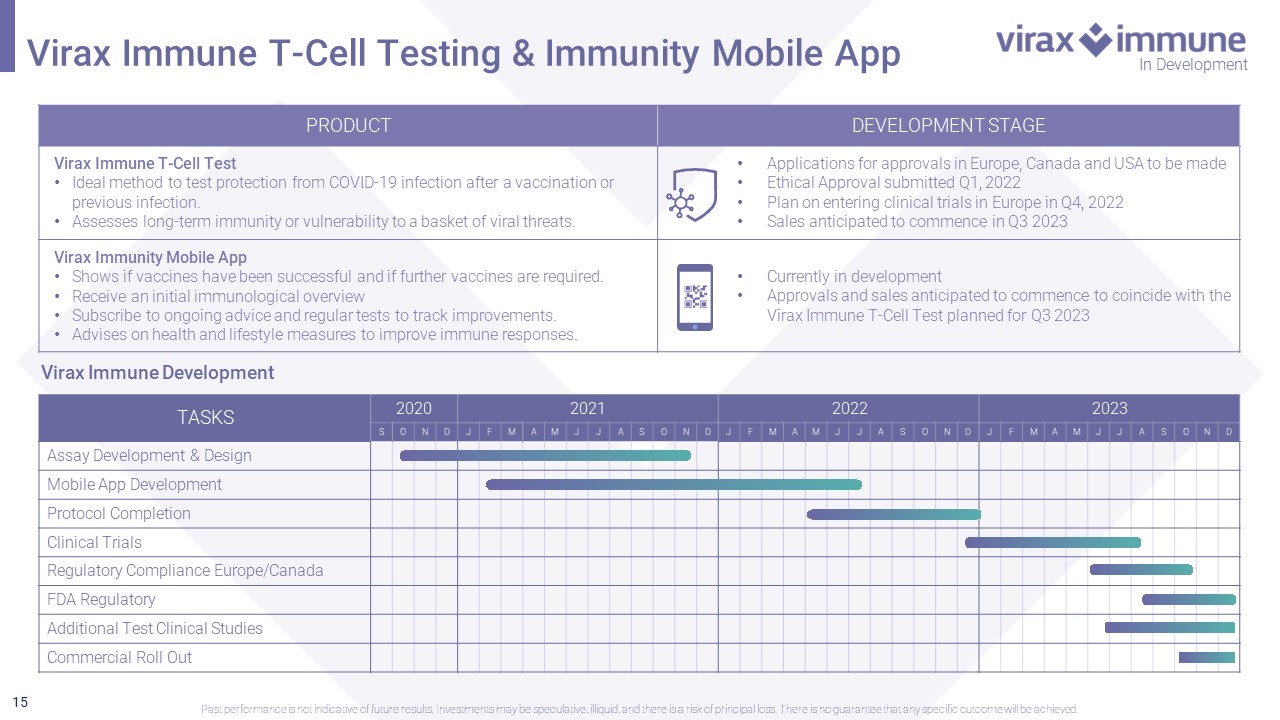
15 In Development Virax Immune T - Cell Testing & Immunity Mobile App Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. PRODUCT DEVELOPMENT STAGE Virax Immune T - Cell Test • Ideal method to test protection from COVID - 19 infection after a vaccination or previous infection. • Assesses long - term immunity or vulnerability to a basket of viral threats. • Applications for approvals in Europe, Canada and USA to be made • Ethical Approval submitted Q1, 2022 • Plan on entering clinical trials in Europe in Q4, 2022 • Sales anticipated to commence in Q3 2023 Virax Immunity Mobile App • Shows if vaccines have been successful and if further vaccines are required. • Receive an initial immunological overview • Subscribe to ongoing advice and regular tests to track improvements. • Advises on health and lifestyle measures to improve immune responses. • Currently in development • Approvals and sales anticipated to commence to coincide with the Virax Immune T - Cell Test planned for Q3 2023 TASKS 2020 2021 2022 2023 S O N D J F M A M J J A S O N D J F M A M J J A S O N D J F M A M J J A S O N D Assay Development & Design Mobile App Development Protocol Completion Clinical Trials Regulatory Compliance Europe/Canada FDA Regulatory Additional Test Clinical Studies Commercial Roll Out Virax Immune Development
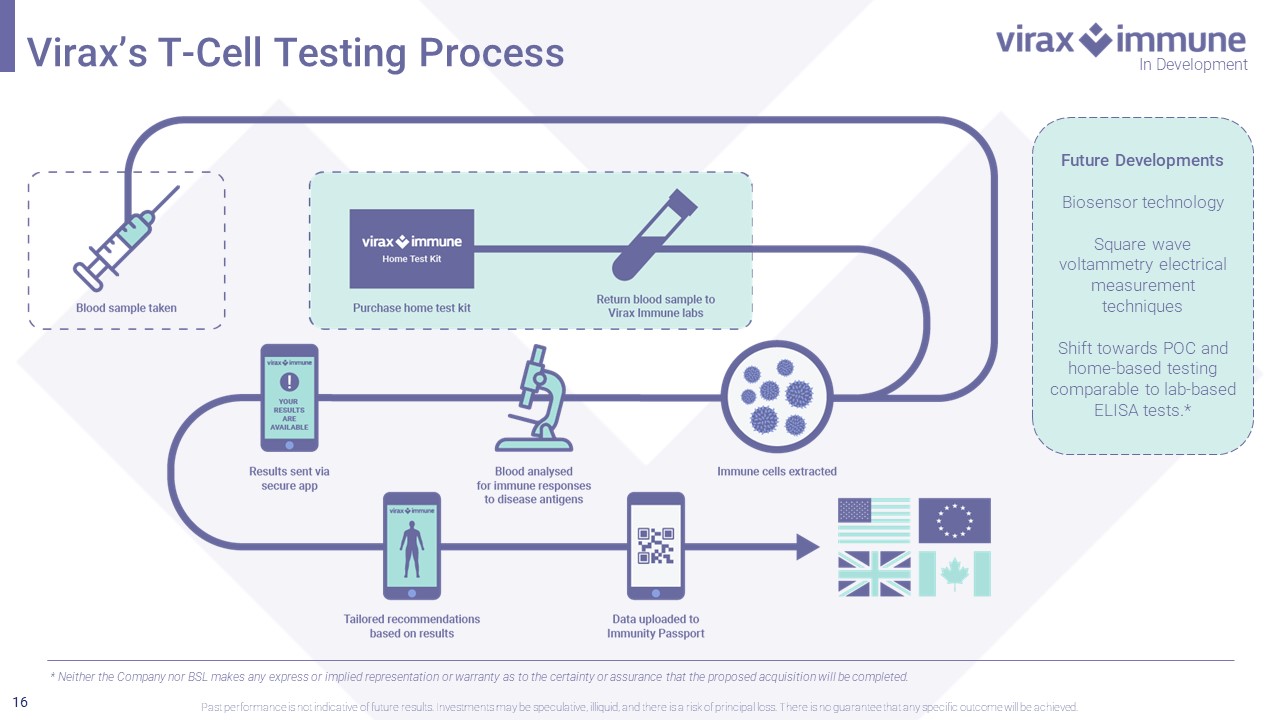
16 Future Developments Biosensor technology Square wave voltammetry electrical measurement techniques Shift towards POC and home - based testing comparable to lab - based ELISA tests.* * Neither the Company nor BSL makes any express or implied representation or warranty as to the certainty or assurance that t he proposed acquisition will be completed. In Development Virax’s T - Cell Testing Process Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.
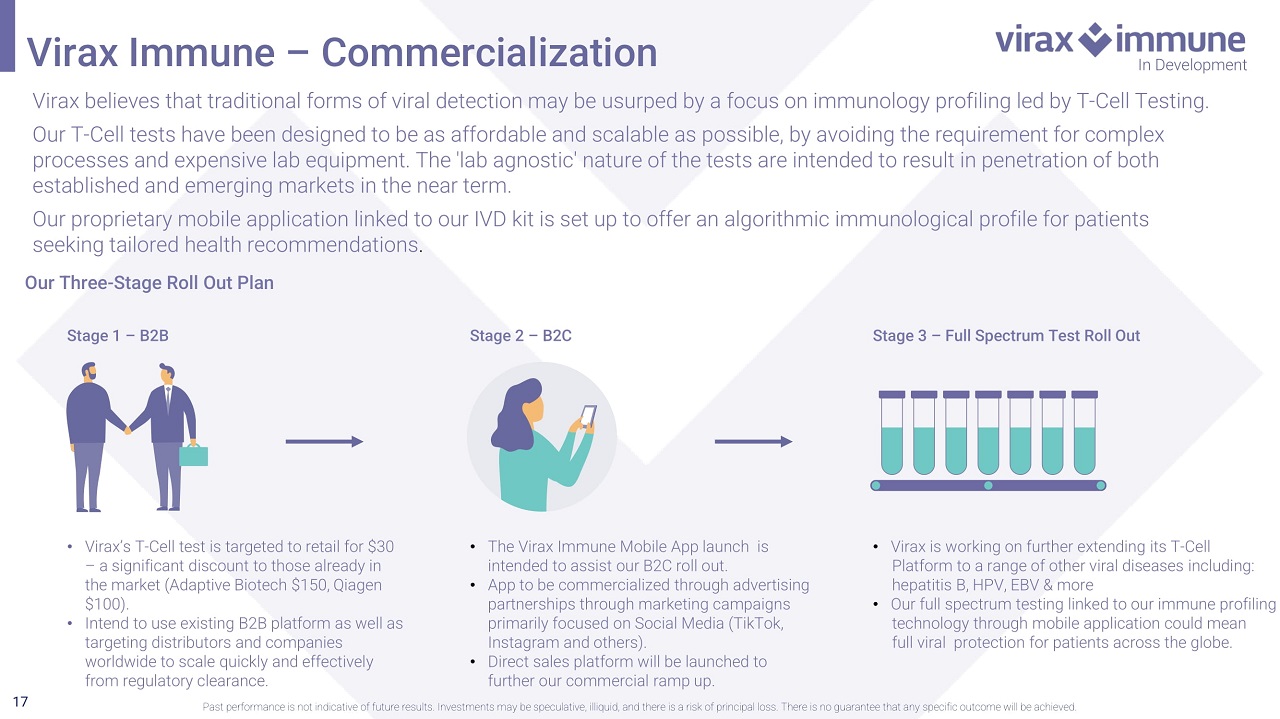
17 Virax Immune – Commercialization In Development Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. Virax believes that traditional forms of viral detection will be usurped by a focus on immunology profiling led by T - Cell Testing. Our T - Cell tests have been designed to be as affordable and scalable as possible, by avoiding the requirement for complex processes and expensive lab equipment. The 'lab agnostic' nature of the tests are intended to result in penetration of both established and emerging markets in the near term. Our proprietary mobile application linked to our IVD kit is set up to offer an algorithmic immunological profile for patients seeking tailored health recommendations . Our Three - Stage Roll Out Plan Stage 1 – B2B • Virax’s T - Cell test is targeted to retail for $30 – a significant discount to those already in the market (Adaptive Biotech $150, Qiagen $100). • Intend to use existing B2B platform as well as targeting distributors and companies worldwide to scale quickly and effectively from regulatory clearance. Stage 2 – B2C • The Virax Immune Mobile App launch is intended to assist our B2C roll out. • App to be commercialized through advertising partnerships through marketing campaigns primarily focused on Social Media (TikTok, Instagram and others). • Direct sales platform will be launched to further our commercial ramp up. Stage 3 – Full Spectrum Test Roll Out • Virax is working on further extending its T - Cell Platform to a range of other viral diseases including: hepatitis B, HPV, EBV & more • Our full spectrum testing linked to our immune profiling technology through mobile application could mean full viral protection for patients across the globe.

18 19. As of 24/05/2022, source: Google Finance COMPETITOR DETAILS VIRAX IMMUNE POINT OF DIFFERENCE Oxford Immunotec is a diagnostics company that currently offers a T - Cell Test for COVID, Cytomegalovirus and Tuberculosis. The Company was recently acquired by Perkin Elmer in January 2021 for $591 million . • We believe that our sensitivity will be higher. • Expected to increase the number of detectable T - cells over the presence of just one cytokine in isolation. A daptive Biotechnologies is a clinical diagnostics company that operates under the premise that data from the immune system is the key to diagnosing and treating diseases. The Company listed on NASDAQ in 2020 and has a market cap of $1.12 billion 1 . • Virax Immune flow cytometry - based test requires less specialized laboratory equipment and reagents than Adaptive Biotech’s testing kits. • Virax’s test kits are being developed to cost significantly less than those offered by Adaptive. • Virax leverages more widely available test devices, reagents and techniques, and sourcing from our factories and supply chain partners in East Asia. TScan Therapeutics is a biopharmaceutical company focused on the development of T - cell receptor (TCR) - engineered T cell therapies in oncology. The Company recently partnered with Qiagen, a NYSE - listed company with a market cap of $68.49 million 1 . • Virax focuses on diagnosis and tailored lifestyle recommendations while TScan is more focused on T - Cell therapeutics. T - Cell Diagnostics Competitor Analysis In Development Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.

COMPETITOR STOCK TICKER NASDAQ: ADPT NYSE: QGEN CURRENT MARKET CAP $1.12 billion $10.44 billion COMPANY DETAILS • Currently own and operate 33 active patents • Completed IPO in 2019. • Not profitable but revenue generating to this day. • Raised $465.69mm in order to get to IPO. • Has had to raise significantly more funding to get to the same stage as Virax . • Strong buy Indicated by analysts. • $36.2 million pre - IPO funding at $185 million post - money valuation. • 548 patents from inception 1986. • Raised $590mm to date. • Strong buy from analysts. 19 Where Could Virax Be In The Future? Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved.

20 James Foster Chief Executive Officer James has been an Asia - focused investment, product development and distribution expert in the medical device and pharmaceutical sector for over 10 years. Cameron Shaw Chief Operations Officer Cameron has a depth of experience in Business Development, Strategy and Sales, having managed successful teams and companies across Asia and The UK. Dr Tomasz George Chief Scientific Officer Tomasz has a PhD in Applied Physiology and Ageing from King’s College London. He has 12 years of experience in developing digital healthcare systems. Mark Ternouth Chief Technical Officer Mark is a graduate of Cambridge University and a Chartered Accountant (ACA - ICAEW). He specialises in transforming businesses through technology. Jason Davis Chief Financial Officer Jason is a licensed certified public accountant and has served as the CFO and Principal Accounting Officer for both Nasdaq and NYSE listed companies. Yair Erez Independent Director Yair is a Partner at Bain & Company, where he works with private equity investors across the investment cycle – deal sourcing, buy side diligence, post acquisition work and exit support, with a focus on the healthcare and life sciences industries. Peggy Gilmour Independent Director Peggy Gilmour is a senior finance, risk management and audit executive with a deep understanding of both US and Canadian regulatory environments. She is current audit & risk committee chair for Nasdaq listed POINT Biopharma. Evan Norton Independent Director Evan is a Managing Partner at Ballast Capital. Previously he served as Director of Abbott Ventures for almost a decade. He also holds an MBA from Northwestern’s Kellogg School of Management where he is an adjunct lecturer. Team – Management & Non - Executive Board Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. Karen Bertoli Advisory Board Member Karen is engaged with numerous innovative and disruptive technology organisations across Finance, Healthcare, eCommerce, and Digital Assets. Pierre Frouin Advisory Board Member With broad experience in the healthcare industry, Pierre founded BioSerenity , an innovative company focused on the use of A.I. for the digital detection of biomarkers.

21 Our Platform is a Disruptive, Innovative Growth Engine Virax Biolabs is positioned to be a growth - oriented biotechnology company with the launch of Virax Immune. We believe our product suite is disruptive, innovative and will gain market share rapidly. Intellectual Property Protection Our Intellectual Property development process is seamless and agile with an in - house science team working alongside Virax’s development partners. Virax has patent protection over it’s T - Cell IVD testing and continues to develop its IP portfolio regularly. Existing Revenue Streams Virax has multiple B2B2C sales channels, an established distribution network, and its own online sales platform. Our high quality and low prices have resulted in distribution in both emerging and developed markets. Investment Highlights Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. Ground Floor IPO O ther biotech companies are burdened with investing into multiple business segments which compete internally for R&D dollars and growth channels. Virax is focused on detection and diagnosis within the IVD market. Investors can get in on the ground floor of a company focused on high - demand, viral - related markets, at an attractive valuation.
22 Office Locations & Contact Past performance is not indicative of future results. Investments may be speculative, illiquid, and there is a risk of princi pal loss. There is no guarantee that any specific outcome will be achieved. Online www.viraxbiolabs.com info@viraxbiolabs.com London Headquarters 20 North Audley Street, London W1K 6LK United Kingdom Singapore Office 531A Upper Cross Street #04 - 98, Singapore, 051531

Thank You www.viraxbiolabs.com